Technology
– Lowering CAPEX and increasing efficiency
The overall objective of the MeSAF project is to fast-track the development and demonstration of a very promising technology that converts raw e-methanol (undistilled) to Sustainable Aviation Fuel (SAF) using a groundbreaking catalyst from Vertimass (US based company).
There are several advantages of the Vertimass alcohol to sustainable aviation fuel technology. Firstly, it does not require distillation of the methanol resulting in lower CAPEX and higher efficiency. Secondly, it only requires one processing step whereas other methanol to aviation fuel technologies, based on existing methanol to gasoline technology, require at least three steps.
LPG & SAF from renewable hydrogen and biogenic waste-CO2
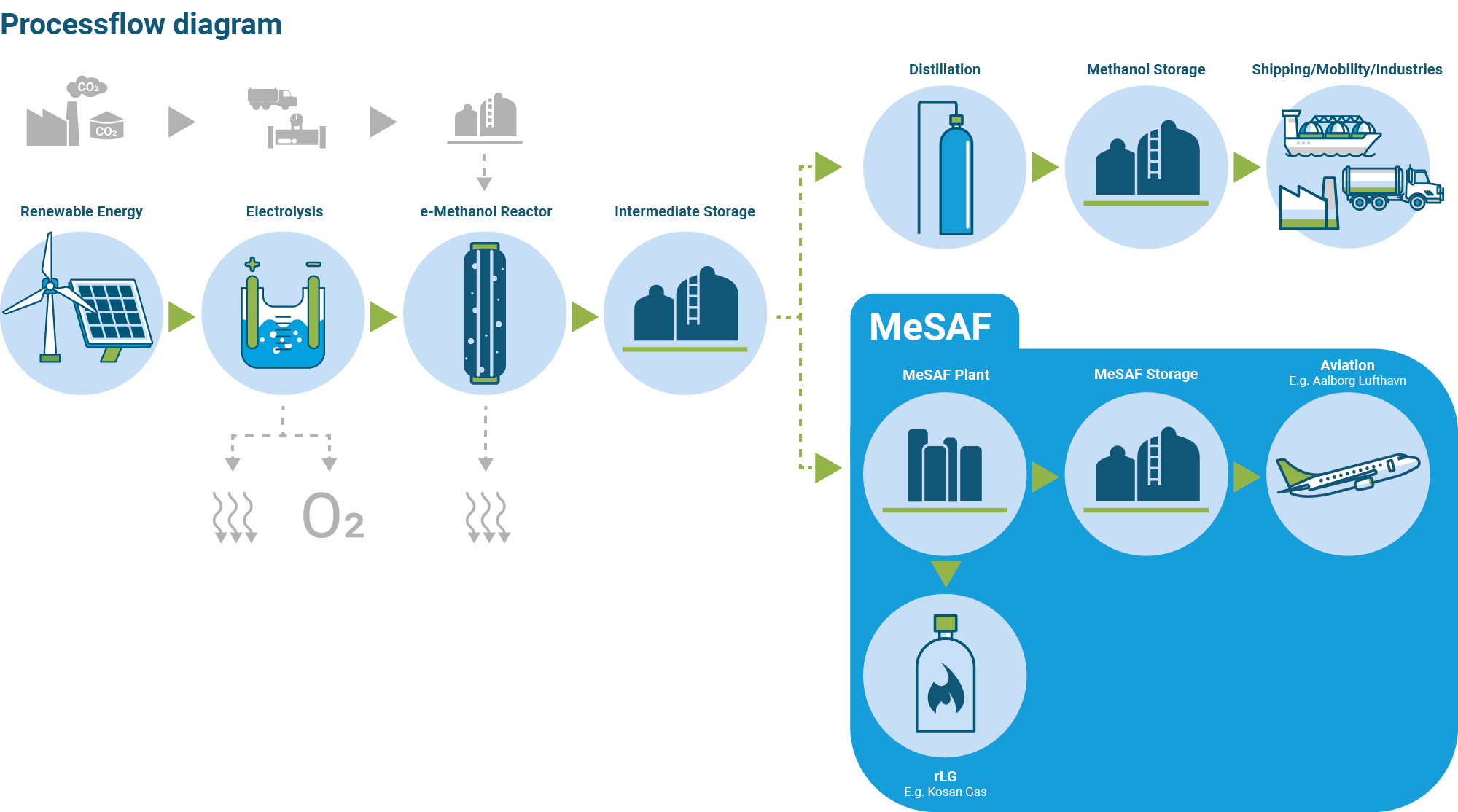
eMethanol is produced by chemically combining CO2 and hydrogen. First, hydrogen is produced in an electrolyzer by using renewable electricity to split water (H20) into hydrogen (H2) and oxygen (O). The hydrogen is converted into methanol in a reactor by catalysis with carbon dioxide (CO2). After the methanol synthesis of the hydrogen and CO2, a raw/undistilled eMethanol product has been produced, containing approx. 2/3 Methanol and 1/3 water.
Once the raw eMethanol is obtained, it can be further processed into 2 different utilizations. Either the raw methanol can be distilled in distillation towers and the water is separated from the methanol to produce either 1. pure eMethanol for a multitude of transport and industry sectors or 2. utilized as feedstod for Vertimass catalyst technology to produce primarily SAF but also a significant amount of byproducts such as LPG and Nafta.
Converting renewables into SAF
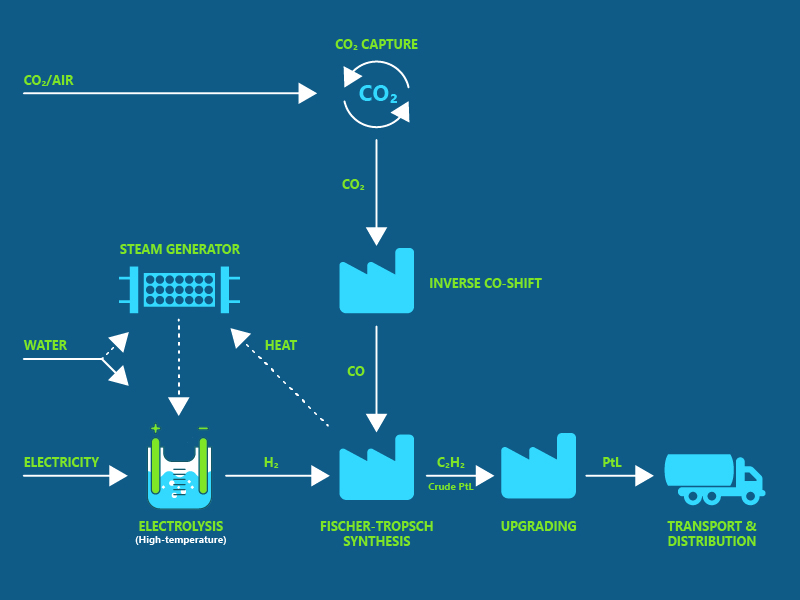
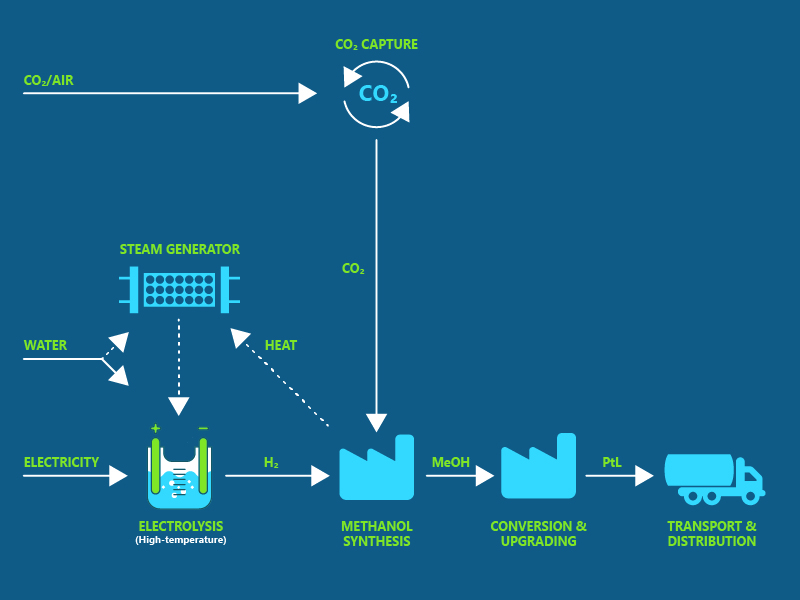
One of the most sustainable and scalable routes to SAF is via green hydrogen and CO2 that is converted to a synthetic fuel either via methanol as an intermediate or by Fischer-Tropsch synthesis.
Slide in illustration right to see the two different process flows.
MeSAF plant takes Power2Met to next level
Power2Met was the first of its kind e-methanol power plant built in Denmark. It received funding by the Danish Energy Technology Development Demonstration (EUPD) for its establishment.
It was built with the purpose of demonstrating production of e-methanol, initiated in 2019, and fully operational since 2021. It operates today at small scale. MeSAF can be considered a step-up version to Power2Met, integrating the Vertimass catalysts for cost-effective, large-scale production to MeSAF and rLG.
At Vertimass, we seek to develop and widely commercialize breakthrough technologies that substantially expand the use of sustainable transportation fuels and chemicals that reduce greenhouse gas emissions and improve energy security and domestic economies. Our novel technology enables lower costs of sustainable fuels and chemicals and it’s great to showcase in the MeSAF project.